NSD3-Short Is an Adaptor Protein that Couples BRD4 to the CHD8 Chromatin Remodeler
Abstract
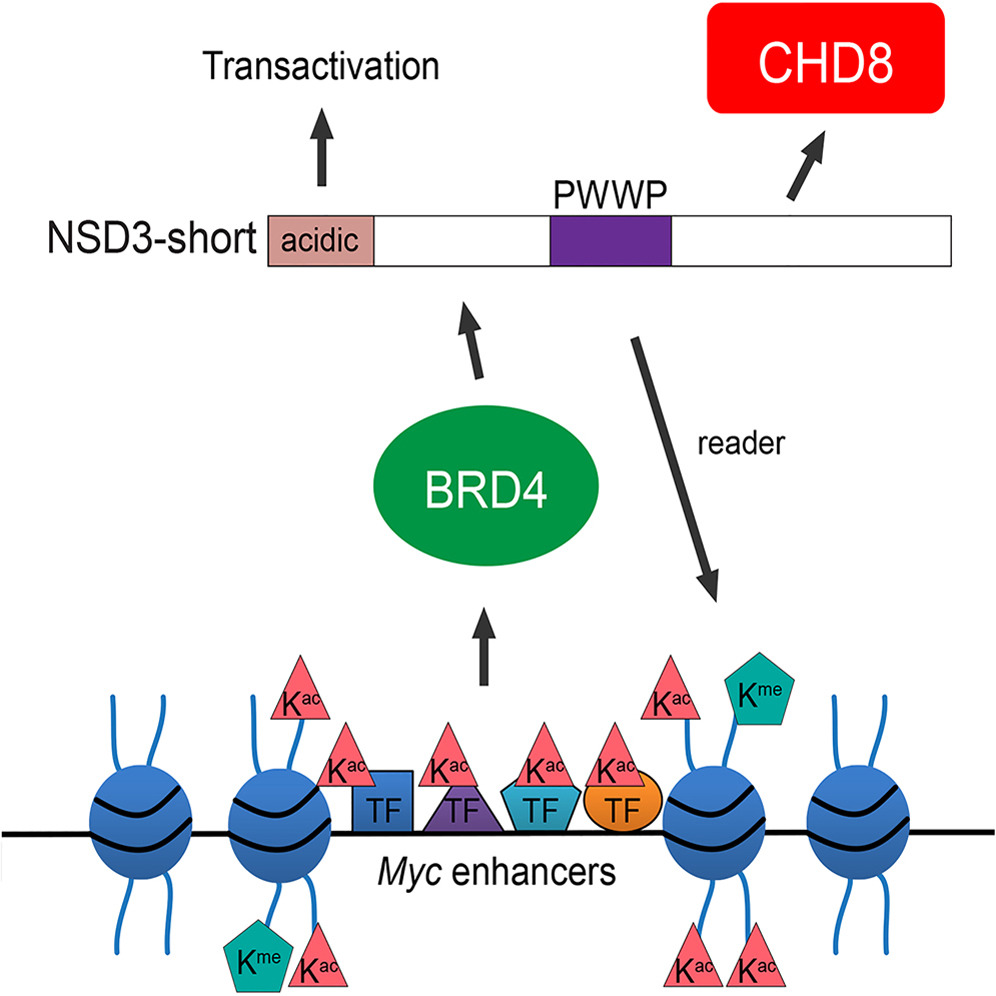 The bromodomain and extraterminal (BET) protein BRD4 is a therapeutic target in acute myeloid leukemia (AML). Here, we demonstrate that the AML maintenance function of BRD4 requires its interaction with NSD3, which belongs to a subfamily of H3K36 methyltransferases. Unexpectedly, AML cells were found to only require a short isoform of NSD3 that lacks the methyltransferase domain. We show that NSD3-short is an adaptor protein that sustains leukemia by linking BRD4 to the CHD8 chromatin remodeler, by using a PWWP chromatin reader module, and by employing an acidic transactivation domain. Genetic targeting of NSD3 or CHD8 mimics the phenotypic and transcriptional effects of BRD4 inhibition. Furthermore, BRD4, NSD3, and CHD8 colocalize across the AML genome, and each is released from super-enhancer regions upon chemical inhibition of BET bromodomains. These findings suggest that BET inhibitors exert therapeutic effects in leukemia by evicting BRD4-NSD3-CHD8 complexes from chromatin to suppress transcription.
The bromodomain and extraterminal (BET) protein BRD4 is a therapeutic target in acute myeloid leukemia (AML). Here, we demonstrate that the AML maintenance function of BRD4 requires its interaction with NSD3, which belongs to a subfamily of H3K36 methyltransferases. Unexpectedly, AML cells were found to only require a short isoform of NSD3 that lacks the methyltransferase domain. We show that NSD3-short is an adaptor protein that sustains leukemia by linking BRD4 to the CHD8 chromatin remodeler, by using a PWWP chromatin reader module, and by employing an acidic transactivation domain. Genetic targeting of NSD3 or CHD8 mimics the phenotypic and transcriptional effects of BRD4 inhibition. Furthermore, BRD4, NSD3, and CHD8 colocalize across the AML genome, and each is released from super-enhancer regions upon chemical inhibition of BET bromodomains. These findings suggest that BET inhibitors exert therapeutic effects in leukemia by evicting BRD4-NSD3-CHD8 complexes from chromatin to suppress transcription.